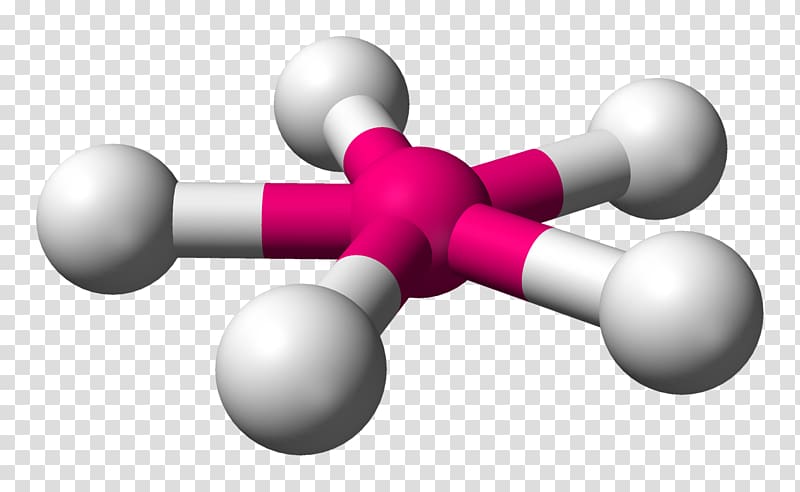
First of all, we will calculate the number of valence electrons for all the atoms present in the molecule.Īs selenium is a group 16 element it consists of 6 electrons in its valence shell while fluorine is a group 17 element therefore, all the fluorine atoms have 7 valence electrons.Let us understand the step by step process for drawing the lewis structure for the SeF4 molecule: the electronic configuration of all the four fluorine atoms as well as the selenium atom is eight. It can be seen in the structure that the octet for all the five atoms bonded to form the SeF4 molecule is satisfied i.e. The Lewis structure for SeF4 is written as: These are also called Electron dot structures or Lewis dot structures. Like any other method of representing molecular structures, it aims at establishing the most stable structure for a molecular with formal charge closest to zero and electronic configuration of valence shell closest to 8 i.e. They introduced the convention of presenting the atoms through their atomic symbols and their valence electrons as the dots surrounding them. Lewis structures were introduced by Gilbert N. This is known as the octet rule.Īs per this rule, all the atoms tend to form chemical bonds with other atoms either by sharing or donating, or accepting electrons from their valence shells in an attempt to complete their octet or acquire eight valence electrons in order to achieve stability. 
Therefore, it is established that an atom of any element becomes stable when it has eight valence electrons. It is already known that all noble gases, except helium, have eight electrons in their outermost shell. Noble gases are considered the most stable elements of the periodic table as they do not easily compromise their properties by reacting with other atoms. 
These electrons actively participate in the chemical bonds that an atom forms with other atoms and are also given away (or taken up) from one atom to another in the case of ionic bonding. the electrons that are present in the valence shell of an atom. The valence electrons are the electrons that are farthest from the nucleus of an atom i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
